- Your Product
- AUTOMATION SOLUTIONS
- CAPABILITIES
- SERVICES
- RESOURCES
- About Us
- Brands & Affiliates
- Quality
- Environment
- Locations
- Careers
- Contact
- Your Product
- AUTOMATION SOLUTIONS
- CAPABILITIES
- SERVICES
- RESOURCES

NEW Platform Finder!
A new tool from ATS to help you find your automation solution FAST! Review the specs and benefits of our configurable platforms – 70% standard, 30% customizable, 100% ATS reliability.

Automation Solutions
We’ll custom design or help you find the right machines for your Assembly, Material Handling, Conveyance, Manufacturing, Vision Testing, Software (IIoT) needs

SERVICES
Our ability to assist you with your project begins with understanding your product and processes. Whatever the stage of your product’s life cycle—product design, product iteration, clinical trials, or full commercial production—ATS Life Sciences Systems can complement your staff with CGMP-experienced consultants, engineers, and skilled trades and service people

Resources
A deeper dive into information and details about LSS solutions, from the experts who work on them every day.
Sign up for Life Sciences News and Updates

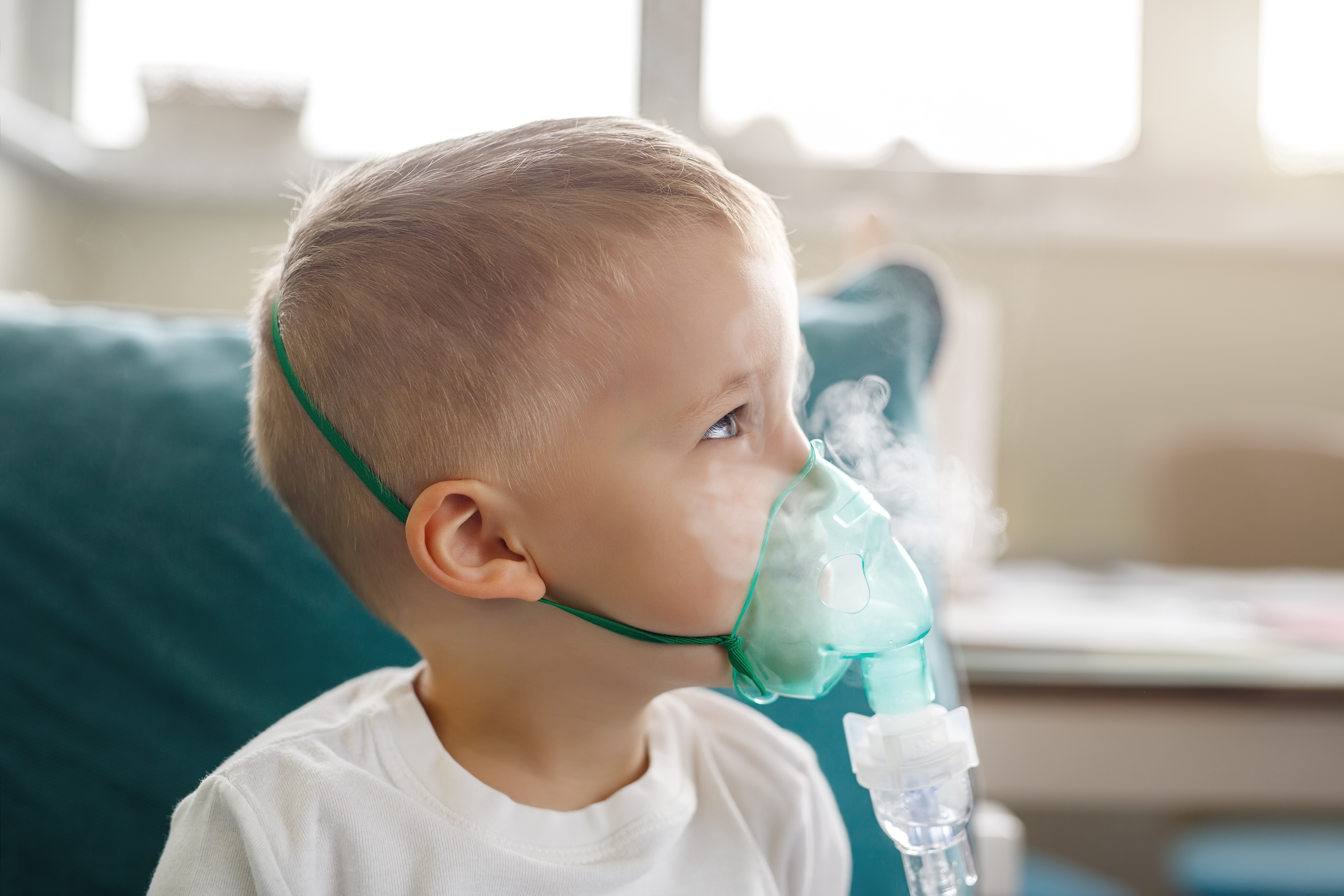
Sub or Final Assembly of Inhalation Devices
INHALER ASSEMBLY
ATS is a leading provider of manufacturing systems for the automated assembly of inhalers. Combination device production of DPIs, MDIs, and other inhalation therapies at full production rates

ATS advanced technologies for inhaled drug delivery device assembly
With extensive expertise in the assembly and packaging of drug delivery devices and pharmaceutical products, ATS can streamline manufacturing processes to manage a broad range of product variants and meet changing market demands.
- Assembly solutions for the inhaled delivery of drugs for respiratory and pulmonary therapies
- Multiple recipe production
- Multiple lines (2-up)
- Smart automated assembly processes and capacity for multiple sub-assemblies
- ISO 5 – 8 levels of cleanroom classifications
AUTOMATE YOUR INHALATION DEVICE ASSEMBLY
ATS automated solutions for inhaler assembly are designed to meet the highest standards of cGMP compliance, FDA regulations, and GAMP 5 guidelines. With proven expertise in pharmaceutical device manufacturing, ATS delivers scalable solutions for:
- Metered Dose Inhalers (MDIs) with integrated counters and valve testing,
- Dry Powder Inhalers (DPIs) with blister strips, annular chambers, and disc formats
- Nebulizers, and
- Soft Mist Inhalers

LOW TO HIGH-SPEED ASSEMBLY
As a trusted leader in medical device assembly, ATS designs modular, scalable solutions tailored to your project’s unique requirements, with the flexibility to adapt to evolving needs.
With a global presence, ATS provides proven expertise in inhaler assembly with precision control for tight tolerances, complex geometries, and rigorous in-process quality controls.
Our modular platforms grow with your product development and expansion, providing end-to-end support from design to commercialization with high-speed production up to 600 parts per minute, depending on the complexity of your assembly.
Why Choose ATS Life Sciences Systems for Injection Device Assembly?

EXPERTISE
Decades of life sciences assembly expertise

Support
Validation support for FDA, cGMP, and GAMP 5 compliance

FLEXIBILITY
Flexible systems for both high-volume and small-batch production
SELECT YOUR INHALER TYPE
Looking for a standardized platform, or ready to explore the potential of a customized inhaler assembly option?
We deliver proven solutions to ramp up your inhaler assembly for fast results, or work with you to develop a fully custom line for multiple devices.
GET IN TOUCH
Briefly tell us about your automation needs and we’ll get back to you.
👋 Looking for something? I'm Gears and am happy to help.






 Contact Us
Contact Us  Subscribe
Subscribe  LinkedIn
LinkedIn  Youtube
Youtube