- Your Product
- AUTOMATION SOLUTIONS
- CAPABILITIES
- SERVICES
- RESOURCES
- About Us
- Brands & Affiliates
- Quality
- Environment
- Locations
- Careers
- Contact
- Your Product
- AUTOMATION SOLUTIONS
- CAPABILITIES
- SERVICES
- RESOURCES

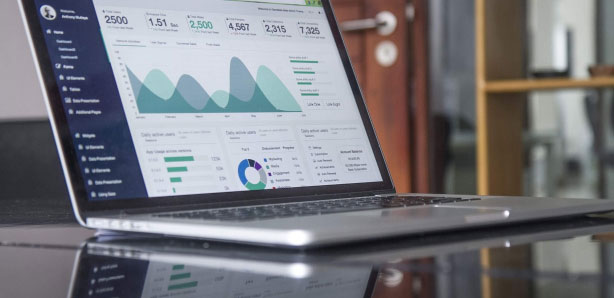
NEW Platform Finder!
A new tool from ATS to help you find your automation solution FAST! Review the specs and benefits of our configurable platforms – 70% standard, 30% customizable, 100% ATS reliability.

Automation Solutions
We’ll custom design or help you find the right machines for your Assembly, Material Handling, Conveyance, Manufacturing, Vision Testing, Software (IIoT) needs

SERVICES
Our ability to assist you with your project begins with understanding your product and processes. Whatever the stage of your product’s life cycle—product design, product iteration, clinical trials, or full commercial production—ATS Life Sciences Systems can complement your staff with CGMP-experienced consultants, engineers, and skilled trades and service people

Resources
A deeper dive into information and details about LSS solutions, from the experts who work on them every day.
Sign up for Life Sciences News and Updates

A successful automation project is more than a machine
QUALITY
Our ability to assist you with your project begins with understanding your product and processes. Whatever the stage of your product’s life cycle—product design, product iteration, clinical trials, or full commercial production—ATS Life Sciences Systems can complement your staff with CGMP-experienced consultants, engineers, and skilled trades and service people
Quality Management
Quality is more than customer satisfaction. It is also a commitment to deliver automation solutions that are compliant with customer and regulatory requirements. This belief is the basis for our quality policy. We demonstrate our commitment to our quality policy every day through the following activities and more.
Environmental Management
Our people are the core of our business; protecting their safety and health and the environment is a fundamental business priority. Our corporate values are key to the ATS Sustainability program, which includes Health and Wellbeing and Safety Culture, where we support our people to foster a safe, positive, and inclusive workplace where everyone is respected and given the opportunity to do their best. We are also a founding member of the Estainium organization.


Certificates
Processes and templates implemented for design reviews, change control, non-compliance issues, requirements traceability, and commissioning and qualification:
Supplier Management and auditing, Internal auditing regimen, In-process and final product inspections
- ISO9001:2015
- Documented history of ISO13485 certification
- Adoption GMP and GDP Practices :




The Highest Standards for International Applications
We have successfully delivered cGMP compliant solutions by exploring appropriate technologies and approaches for automating different processes. Certifications, continuous improvement, and innovation mean we deliver the best possible quality.
CONTACT US
GET IN TOUCH
Briefly tell us about your automation needs and we’ll get back to you.
👋 Looking for something? I'm Gears and am happy to help.



 Contact Us
Contact Us  Subscribe
Subscribe  LinkedIn
LinkedIn  Youtube
Youtube